The Most Robust Platform for Compliance Risk Assessment & Mitigation
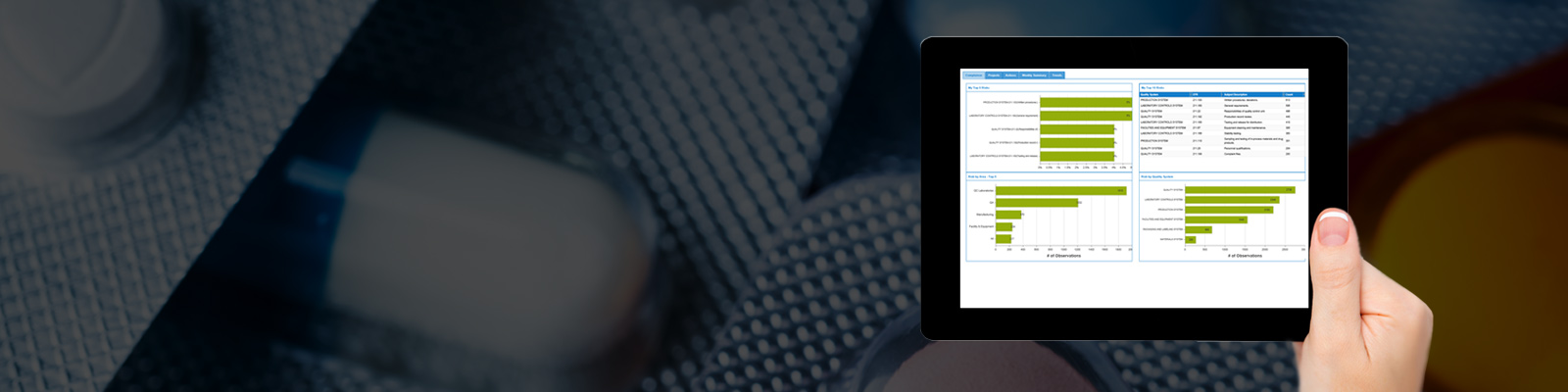
FDAAWARE is an innovative service for FDA’s regulated industries, projecting compliance risks by leveraging current and historical 483s, recalls and warning letters A company profile is used in conjunction with an advanced Projection Algorithm to assess specific sites and overall company compliance related risks, providing the site’s team with potential compliance deficiencies sorted in the order of importance, to avoid the costly consequences of incompliance. Trending reports are pushed to the compliance team, centralizing their focus on up-to-date FDA trends observed in their country, specific industry, region etc., creating a continuous improvement process. FDAAWARE is fully integrated with FDA CFRs for Drugs, Biologics and Devices creating a fully aligned risk management platform.
With over 25 different reports, you can increase your awareness to various KEY industry trends that could affect your next inspection.
We have a team of professionals that constantly monitor new information about the life science industries; they are in contact with the FDA regularly and obtain the most up-to-date information about inspections, 483s, recalls, warning letters and more. Our DB contains over 55,000 Inspections since 2008, more than 15,000 483s dated from 2006, and Recall Information since 2012. Our professional team updates ALL of the above information regularly. We also have Information about more than 2,500 Inspectors and their statistics (current and historical).
Contact us to learn more: info@fdaaware.com

Cloud Services
Our solutions are hosted on Amazon Cloud, one of the most flexible, scalable and reliable cloud providers

Support
Our technical support is available to help you troubleshoot any issue and address challenges post implementation

Services
Implementation support, integration and post go-live help you achieve operational excellence

Implementation
Go-Live in less than a week with our compliance partner
WHAT OUR USERS SAY
LATEST NEWS

cResults completed 5 sites implementation for one of the largest pharmaceutical company

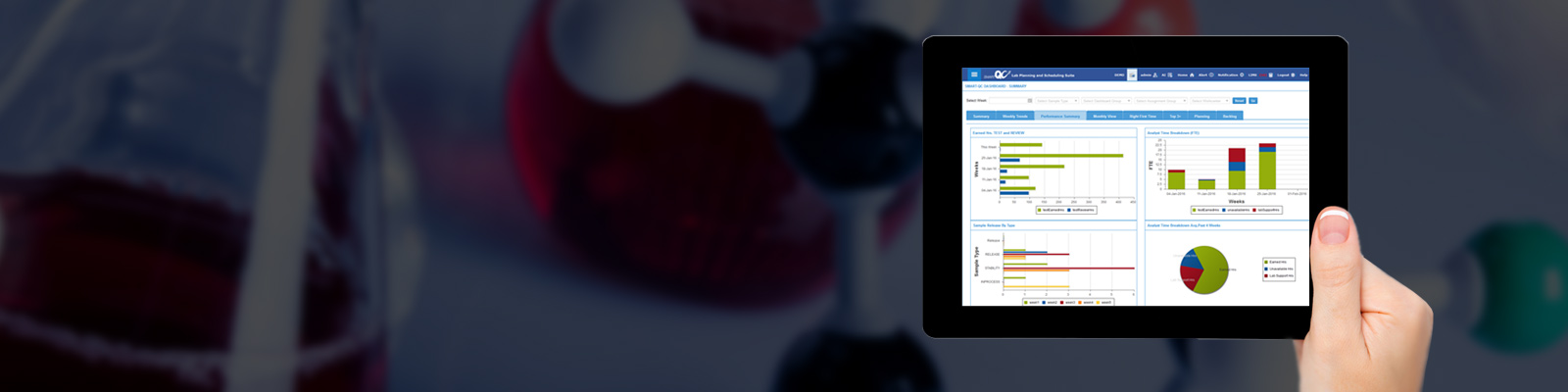
cResults has introduced a performance management module that provides its user to identify any issues related to testing, review, projects and unavailable event in minutes ALL in ONE STOP SHOP display

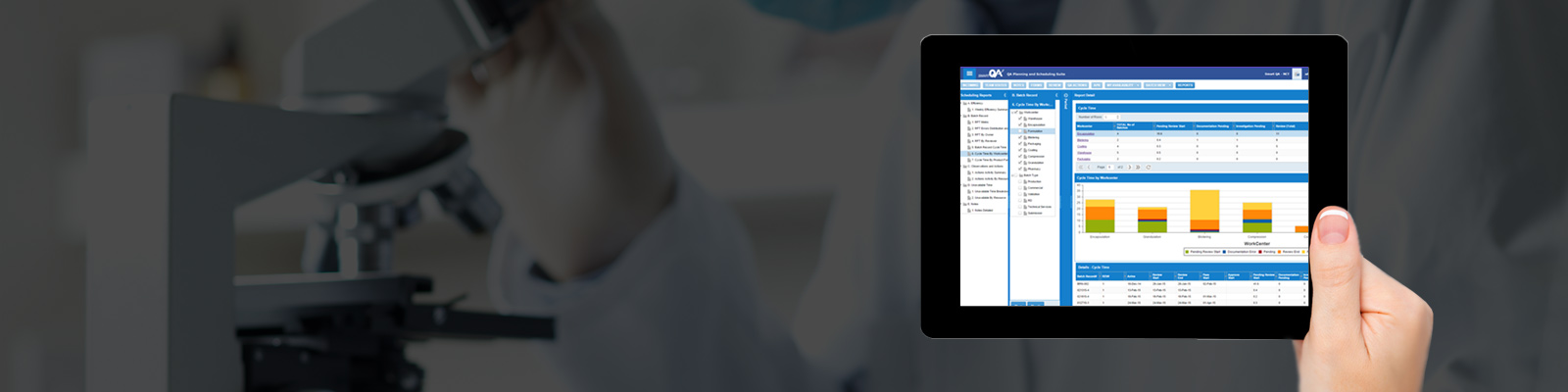
Smart-QA is now integrated with TrackWise and enable to manage the time associated with quality events by QA team

cResults has introduced a performance management module that provides its user to identify any issues related to testing, review, projects and unavailable event in minutes ALL in ONE STOP SHOP display